Mechanics and geneticsOlivier Cochet-Escartin, Hélène Delanoë-Ayari, Sylvain Monnier, Charlotte Rivière We study how the physical and mechanical environment influences the molecular regulations at play in living systems. This type of mechano-genetic coupling is increasingly recognized as an important type of regulation in a variety of biological contexts. We mainly question the role of mechano-genetic feedbacks in three different situations:
Such studies require the control and observation of both the physical environment and the molecular dynamics at play. We therefore combine techniques from microfabrication and soft matter physics with cellular and molecular biology to observe and quantify, in vivo, these two aspects. The goal is to achieve comprehensive models of these important biological processes explicitly taking into account the interplay of mechanics and genetics as a central regulatory element of these processes.
Funding: Ligue régionale contre le cancer, Plan Cancer, Institut François Rabelais, Fondation pour la Recherche Médicale, Institut Lumière Matière (iLM), Fondation de Recherche Andre Marie Ampère (FRAMA)
Collaborations: Centre de Recherche contre le Cancer de Lyon, LiPhy Grenoble, Laboratoire de Régénération et Neurogenèse Adulte (Université de Genève), Porto Alegre (Brasil), Laboratoire de Biologie Moléculaire et Cellulaire (ENS Lyon).
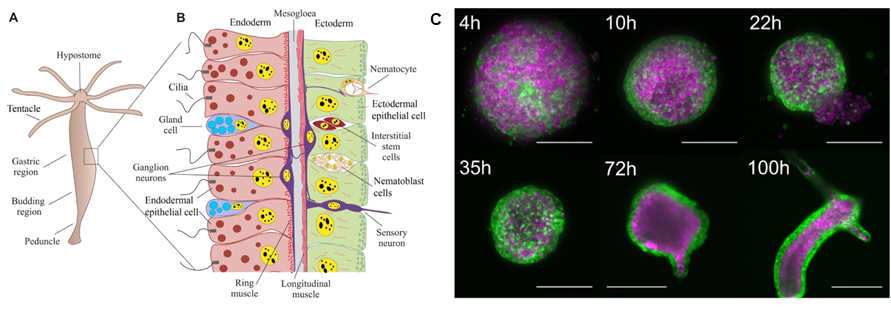
Regeneration of Hydra:We study the role of mechanical signals in the patterning of Hydra vulgaris. This organism is well known for its regenerative capacities allowing to trigger pattern formation at will through amputations or the preparation of cellular aggregates (Fig). We then ask how externally applying mechanical perturbations (pressure, shear) on a regenerating sample affects the regenerative process at the genetic, cellular and organism scale. |
 |
|
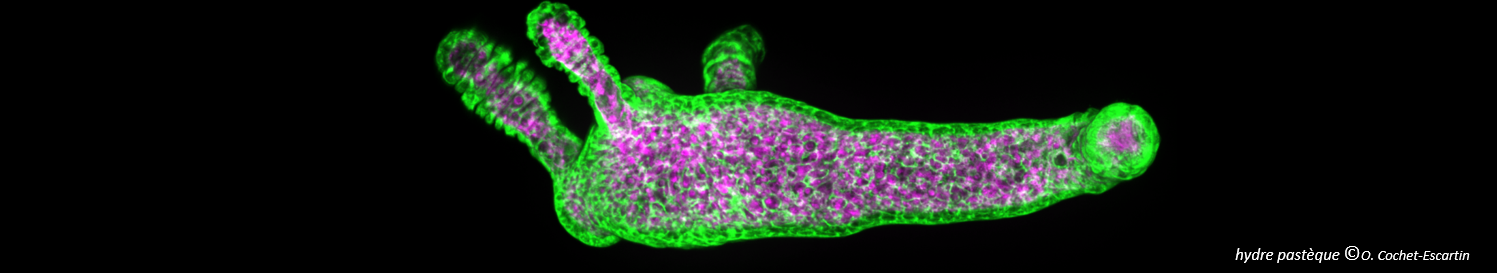
Legend: A-B from (Technau and Steele 2011), C from (Cochet-Escartin et al. 2017). A: Schematic representation of an adult Hydra and its different organs. B: Structure of Hydra at the cellular scale, the two epithelial layers are shown in red and green respectively. C: Timelapse images of Hydra regeneration from an aggregate of cells expressing GFP in the ectoderm (shown in green) and DsRed2 in the endoderm (shown in purple). Scale: 200 mm up to 72h, 500 mm at 100h. |
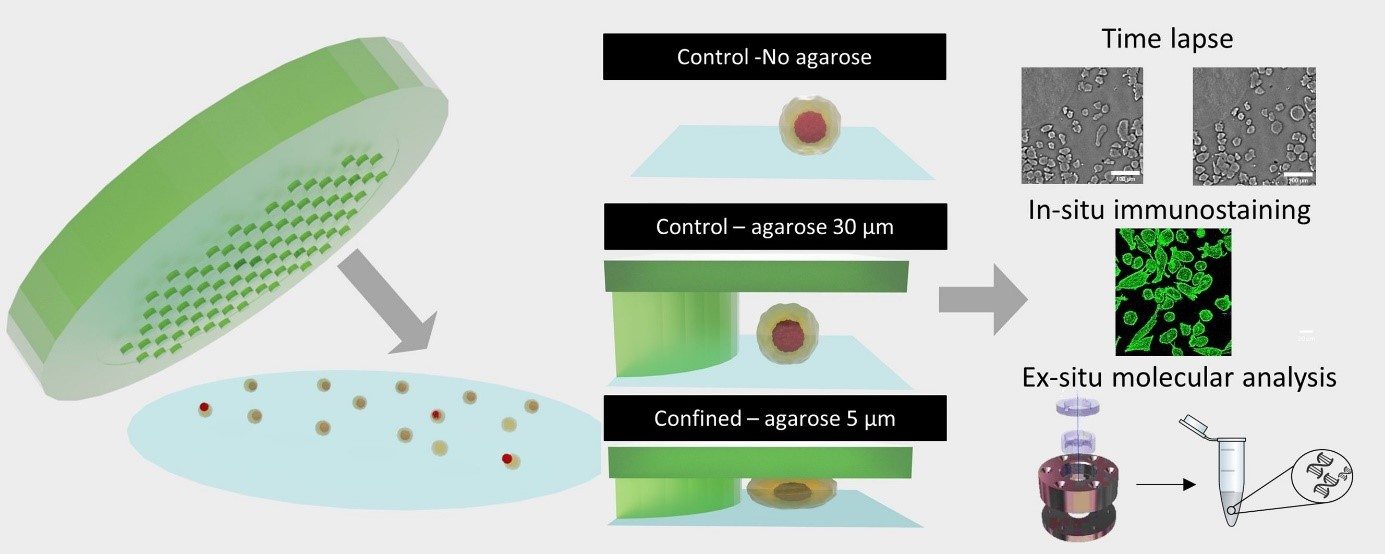
| To do so, we use an interdisciplinary approach combining techniques from molecular biology to observe the dynamics of key patterning genes, soft matter physics to develop force sensors, microfluidics to apply mechanical perturbations (Video 1) and 3d microscopy to observe the entire regenerating sample. |
 |
Regenerating Hydra tissue piece under mechanical perturbations in a microfluidic device. |
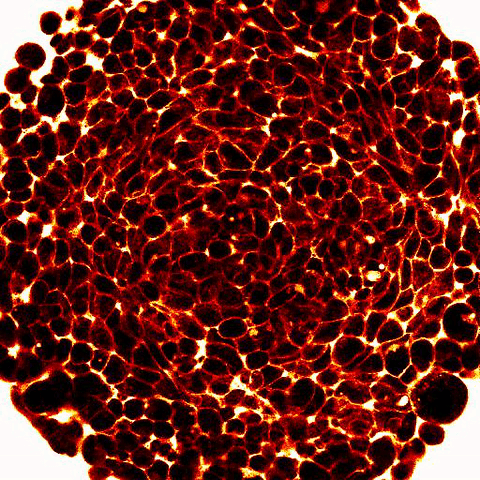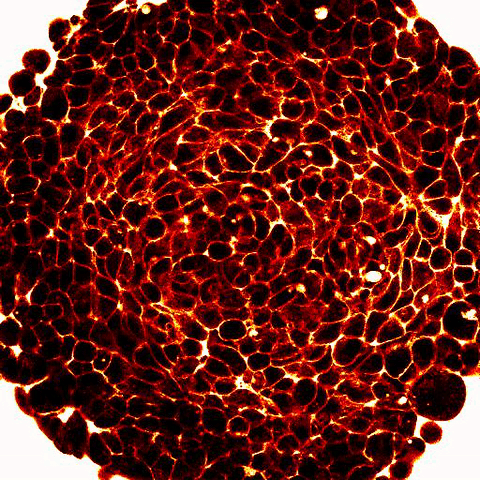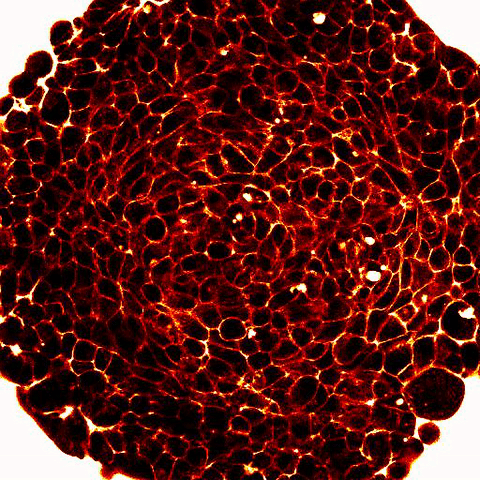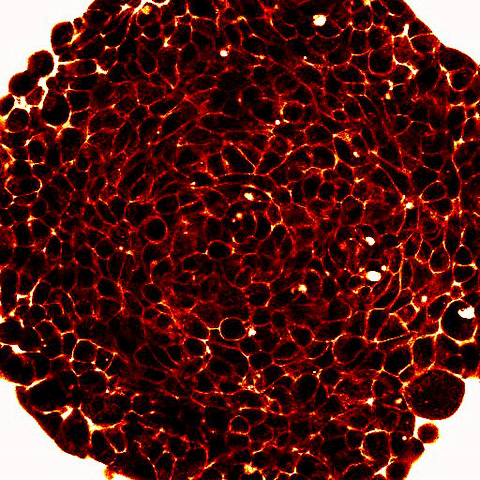 |
Legend: 2-photon imaging of spheroid compression submitted to osmo-mechanical pressure within a microfluidic device. Fluorescence (white and red) labels the space between the cells (dark) and show compression of the intercellular space. |
Key publications:Cochet-Escartin, O., Locke, T. T., Shi, W. H., Steele, R. E., & Collins, E. M. S. (2017). Physical mechanisms driving cell sorting in Hydra. Biophysical journal, 113(12), 2827-2841. |