Physics of Cancer
Investigators: Charlotte Rivière, Hélène Delanoe-Ayari, Jean-Paul Rieu
Cancer Stem cells (CSC) niche
Cell Growth and Signaling analysis in response to mechanical constraints
It has been shown that cell behavior is modified under confinement. However, so far the confined walls were made of rigid material, which cannot be deformed by the cells. What happens if the walls are soft enough to be deformed by the confined cells? We want to address this question using “soft microfluidics” approach. We are currently analyzing the effect on cell growth for MultiCellular Tumor Spheroid Model (Fig1. A) and at the single cell level for Cancer Stem Cells (Fig1. B).
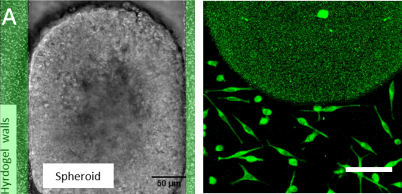
Figure 1: Soft Confinement:
(A) MultiCellular Tumor Spheroid confined in a between 2 hydrogel walls where beads are embedded in order to measure Traction forces exerted by the Spheroid during its growth.
(B) Cancer Stem cells confined in a soft confiner system (h=3.5 µm) thanks to pillars made of hydrogels.
Invasion
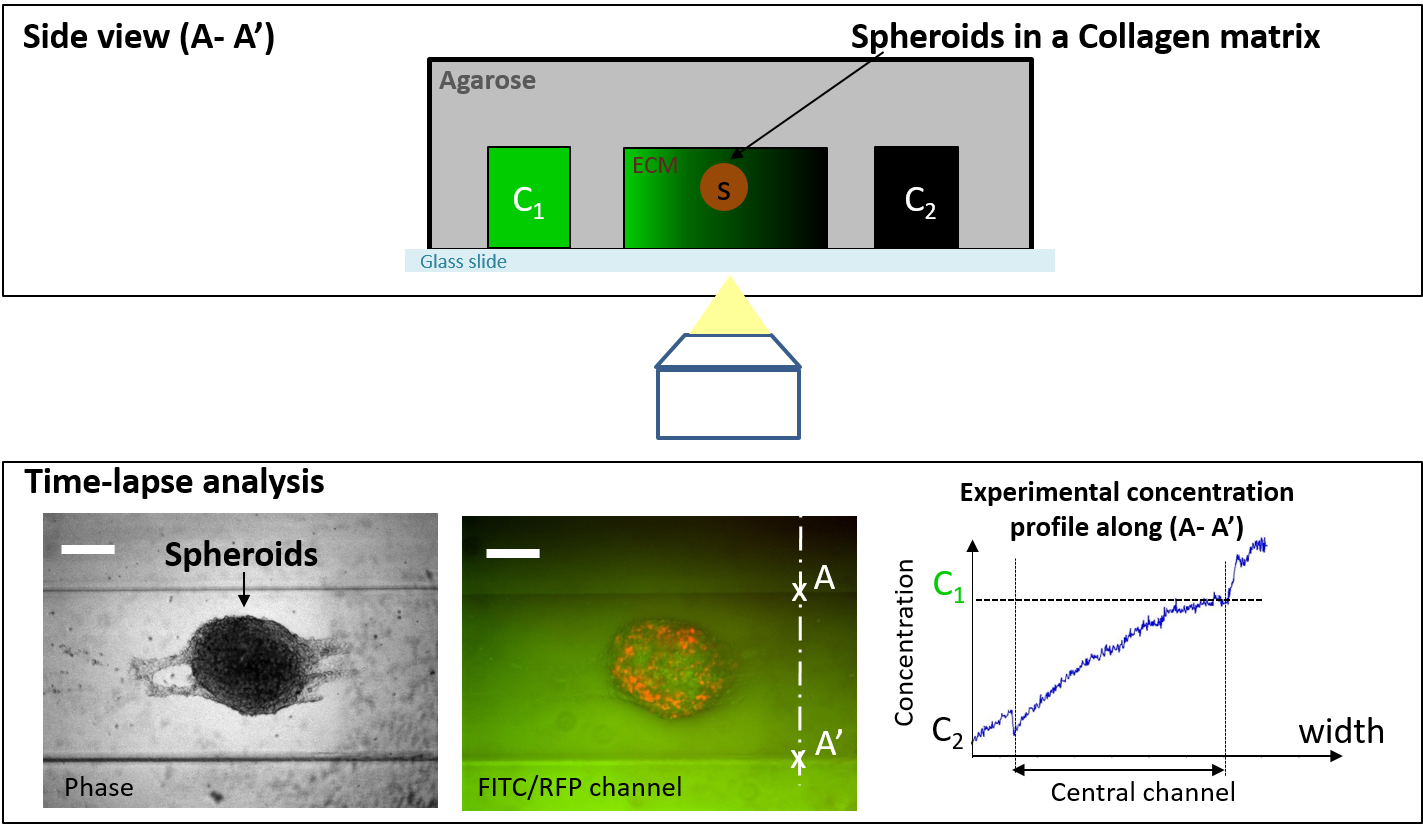
Microfluidic-based system to create 3D in-vivo tumor like environment for drug screening
Many drug candidates for cancer treatment show potential when examined in-vitro but fail in clinical trials. This failure may stem at least in part from the use of conventional in-vitro systems that fail to replicate the physiological microenvironment in humans as well as the lack of cell-phenotypic measurements. We propose here two major improvements for in vitro tests: (1) usage of a 3D micro-environment for cells (the Extra Cellular Matrix (ECM) proteins and fibers, together with gradients of soluble factors secreted by cells), (2) the perfusion of drugs at distance from the biological samples.
we are currently quantitatively analysing cancer cell drug-resistance in in-vivo like environment by implementing an agarose-based microfluidic device (Fig. 2) designed to quantitatively and dynamically assess cellular behaviour and the secretion of various interleukins from (i) 3D multicellular tumour spheroids (MCTS), and (ii) biopsies.
Extravasation Biomimetic
Microsystem to unravel cell capacity to deform and pass through pores of defined geometry and stiffness
We propose to elucidate the cellular mechanisms underlying one critical step in tumor progression: the endothelial transmigration. Indeed, when entering (intravasation) and exiting (extravasation) the vascular vessels, cancer cells need to transmigrate across the endothelial layer of the blood vessel walls which stiffness and geometric confinement creates serious challenges to the cancer cells, requiring them to sustain drastic deformations.
We are currently studying in vitro the extravasation of colorectal cancer cell lines with different metastatic characteristics in microsystems mimicking blood vessels interstices. To do so microfluidic devices implementing networks of micro-pillars and micro-channels were developed (Figure 3). Two main aspects are varied in order to model the in vivo environment associated with the endothelial barrier: the pore geometry and the pore stiffness.
To quantify cell ability to extravasate, several parameters are investigated such as typical deformation undergone by cells, trajectories, transit time and minimum pore-size that the cells are able to cross.
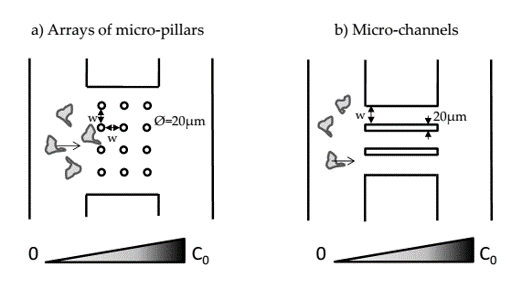
Figure 3: Microsystems for cancer cell migration analysis:
a) Configuration with arrays of micro-pillars. b) Configuration with micro-channels.